2 hydrogen 1 oxygen6/19/2023  
However, the two hydrogen atoms are both on the same side of the oxygen atom so that the positively charged nuclei of the hydrogen atoms are left exposed, so to speak, leaving that end of the water molecule with a weak positive charge. Each hydrogen atom thinks it has two electrons, and the oxygen atom thinks that it has 8 outer electrons. So, oxygen shares one of its outer electrons with each of two hydrogen atoms, and each of the two hydrogen atoms shares it's one and only electron with oxygen. They can get out of this predicament if they agree to share electrons (a sort of an energy "treaty"). Hydrogen and oxygen would like to have stable electron configurations but do not as individual atoms. To balance the negative charge of 8 (2+6) electrons, the oxygen nucleus has 8 protons. The outer ring, on the other hand, has 6 electrons but it would like to have 2 more because, in the second electron ring, 8 electrons is the stable configuration. Oxygen, on the other hand, has two electron rings with an inner ring having 2 electrons, which is cool because that is a stable configuration. 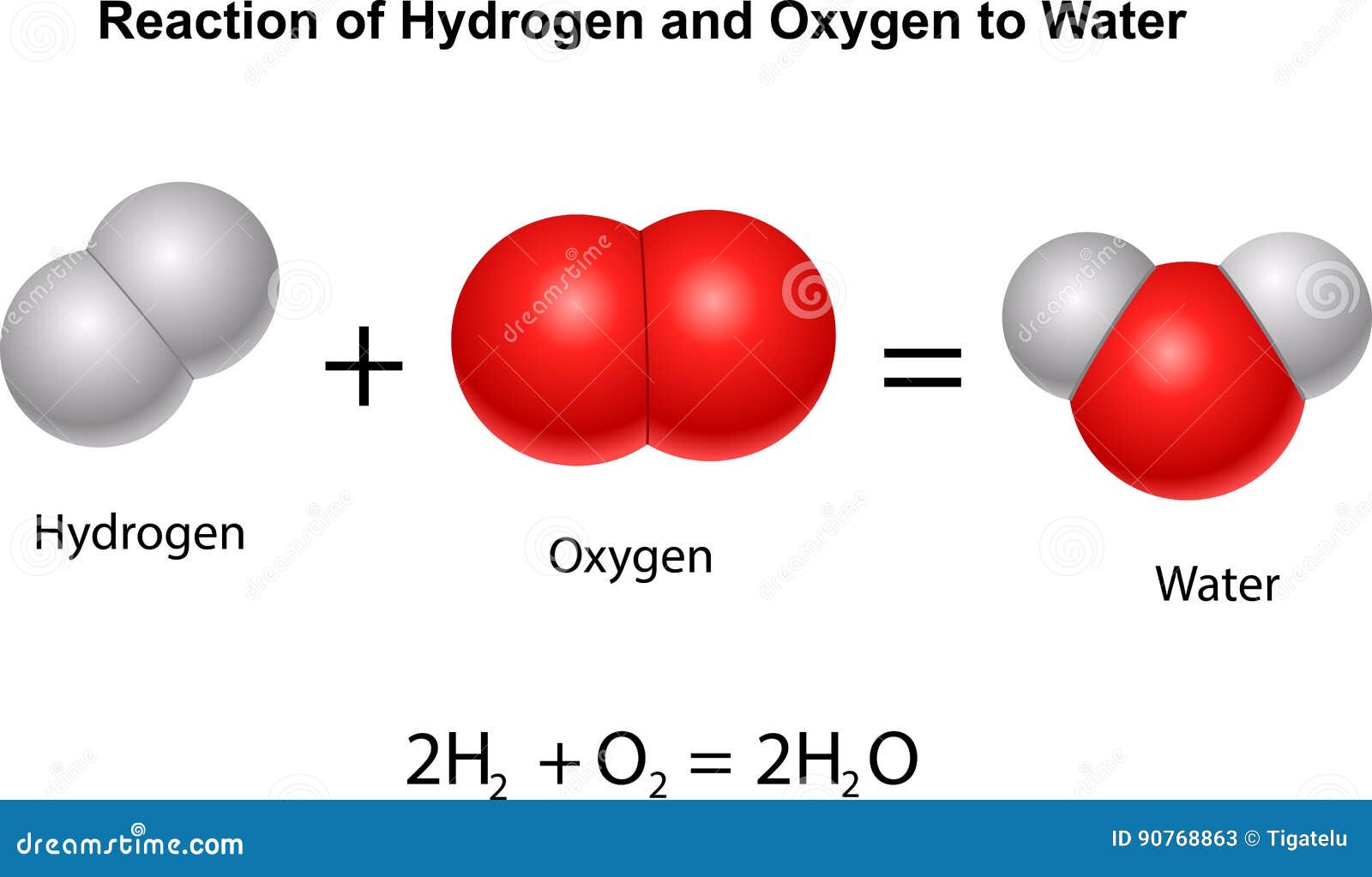
The electron ring of hydrogen would actually prefer to possess two electrons to create a stable configuration. The negative charge of the electron is balanced by the positive charge of one proton in the hydrogen nucleus. The one and only electron ring around the nucleus of each hydrogen atom has only one electron. A molecule of water is composed of two atoms of hydrogen and one atom of oxygen. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
